Chlorella is a single-cell freshwater alga which forms part of a number of microalgae which are cultivated on an industrial scale, and have potential for the production of energy-rich molecules. Researchers at the CEA (French Atomic Energy Commission), the CNRS (French National Centre for Scientific Research), the ESRF, INSERM (French National Institute of Health and Medical Research) and the Universities of Aix-Marseille, Grenoble Alps and Paris-Sud have discovered an enzyme in this algae which allows it to convert certain of its constituent fatty acids into hydrocarbons using light energy only. According to the authors: “This is a major advance in the identification of biological mechanisms which permit the conversion of fatty acids in cells into hydrocarbons, and opens up a new option for the synthesis of hydrocarbons by micro-organisms on an industrial scale”.
In this study, published in Science, researchers from the Institute of Biosciences and Biotechnologies of Aix-Marseille (CEA/CNRS/University of Aix-Marseille), have been able to identify this key enzyme for the synthesis of hydrocarbons , by following its activity and determining a list of potential candidate proteins using a proteomic analysis conducted at the laboratory of Large scale biology (CEA/INSERM/University of Grenoble Alps). The expression in the bacterium E. coli of the encoding gene for the main candidate protein has shown evidence of the production of hydrocarbons, thereby demonstrating that this enzyme was both necessary and sufficient for hydrocarbon synthesis. Characteristic analysis of the pure enzyme revealed that it was capable of splitting a fatty acid into a hydrocarbon molecule and a CO2 molecule, and that this activity required light (Figure 1).
Fatty acid Hydrocarbon
Figure 1. Diagram of the reaction catalyzed by FAP. In a single step, it converts a fatty acid into a hydrocarbon by removing the carboxyl group from the carbon chain (decarboxylation reaction). In the absence of light, the enzyme is inactive.
Researchers have also demonstrated that a co-factor present in the enzyme permits the capture of blue light. The three-dimensional structure of the enzyme (Figure 2), determined by X-ray diffraction analysis conducted on the fully-automated "MASSIF-1" beamline of the European Synchrotron Radiation Facility (ESRF, Grenoble), and kinetic absorption spectroscopy analyses conducted at the Institute of Integrative Cell Biology (CEA/CNRS/University of Paris-Sud), have allowed the formulation of a model for the mechanism of the enzyme. The fatty acid is positioned in a hydrophobic tunnel, at the end of which the co-factor is located. The latter, upon excitation by blue light, removes an electron from the carboxyl group of the fatty acid, thereby resulting in spontaneous decarboxylation to form a hydrocarbon molecule.
The discovery of this enzyme, named FAP (an English acronym for "Fatty Acid Photodecarboxylase") is of major and fundamental interest, on the grounds that, to date, only four biocatalysts capable of exploiting light energy (photoenzymes) have been discovered. FAP is at least ten times more rapid than the best-known enzyme for hydrocarbon synthesis, and employs light, thus potentially providing a highly effective biotechnological tool for the synthesis of hydrocarbons, either by the in vitro conversion of oils, or by the in vivo conversion of the membrane fatty acids of bacteria, yeasts or, ideally, microalgae.
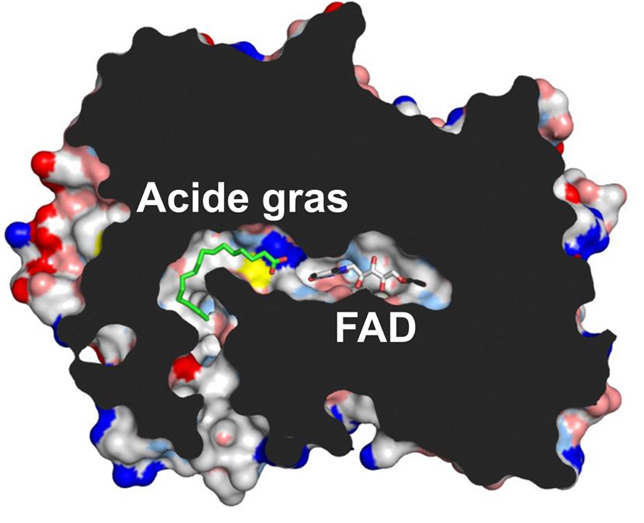
Acid gras = Fatty acid
Figure 2. Slice through the three-dimensional structure of FAP. The flavin-derived co-factor (FAD) is situated at the end of a hydrophobic tunnel, where the fatty acid is fixed (in green). The plane section is indicated in black. © CEA-Biam/Pascal Arnoux.
Reference: D. Sorigué, B. Légeret, S. Cuiné, S. Blangy, S. Moulin, E. Billon, P. Richaud, S. Brugière, Y. Couté, D. Nurizzo, P. Müller, K. Brettel, D. Pignol, P. Arnoux, Y. Li-Beisson, G. Peltier, F. Beisson. An algal photoenzyme converts fatty acids to hydrocarbons.
From fossil fuel to "bio" fuel
At present, hydrocarbons are essentially derived from mineral oil; however, the most easily-accessible fossil fuel reserves are becoming progressively depleted, while the extraction of non-conventional oils is increasingly expensive, in environmental terms.
The production of bio-sourced hydrocarbons employs atmospheric CO2 as a carbon source, and would permit the restriction of the bulk discharge into the atmosphere of carbon stored in the subsurface. Moreover, hydrocarbons are difficult to replace in chemistry, or as fuels in the aeronautical industry. In the interests of limiting the use of fossil fuels, the production of hydrocarbons from living organisms is therefore a key biotechnological objective.
[1] Institute of Biosciences and Biotechnologies of Aix-Marseille (BIAM in Cadarache; CEA/CNRS/University of Aix-Marseille), in collaboration with the Institute of Integrative Cell Biology (I2BC in Saclay; CEA/CNRS/University of Paris-Sud), the Large-scale Biological Laboratory (BGE in Grenoble; CEA/INSERM/University of Grenoble Alps) and the European Synchrotron Radiation Facility (ESRF) in Grenoble.