Diapositive précédente
Diapositive suivante
Vous êtes ici : Accueil Cité des Énergies > ACCUEIL BIAM
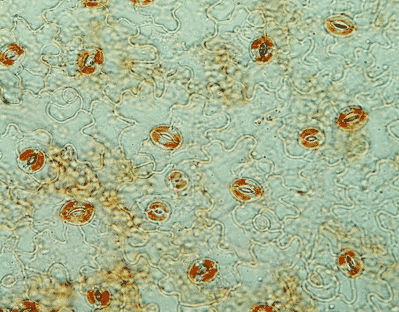
Institut de Biosciences et biotechnologies d'Aix-Marseille
Actualités
Faits marquants
Équipes et thèmes de recherche
Agenda du BIAM
Congrès
Rencontres interdisciplinaires
European Institute of Marine Sciences in Plouzané
Congrès
Le Pharo, Marseille, France
29-05-2024
Conférence
Congrès
Rouen, France
Congrès
Le Pharo, Marseille, France
05-04-2024
Congrès
Rencontres interdisciplinaires
Marseille, Faculté de pharmacie de la Timone, France
08-03-2024
Exposition
Place Général de Gaulle 13001 Marseille
Nos tutelles