A closer look at how the brain works
Upon entering via the galleria - the name given to NeuroSpin’s lobby - visitors are immediately drawn to the sine waves decorating the wall and six archways. There are six of them, like the number of MRIs belonging to this research centre for innovation in neuroimaging at the Frédéric Joliot Life Sciences Institute on the CEA site of Paris-Saclay. “We have three preclinical MRIs which are used on animals, in addition to three clinical MRIs with power levels of 3, 7 and 11.7 Tesla. The last was developed during the Iseult project and it is the only one of its kind. We are currently the most advanced centre worldwide from a technical perspective”, stated Cyril Poupon, the Deputy-Director of the BAOBAB unit and technical coordinator of NeuroSpin’s major facilities. These extremely high-field devices, whether the 7T MRI or the pending 11.7T MRI, are used to collect brain images at very high resolutions. Together with the multidisciplinary teams at NeuroSpin specialised in fields like electronics, IT, image processing and physics with ultra-high magnetic fields, these devices are designed to precisely understand how the brain works, whether in 'normal’ or pathological conditions. Psychiatric diseases such as bipolar disorder, schizophrenia and autistic problems are among the pathologies investigated in this centre.

The galleria - the name given to NeuroSpin’s entrance hall © CEA
Better understanding psychiatric diseases
According to Professor Josselin Houenou, head of the psychiatric team in NeuroSpin’s Uniact unit, “the objective is to better understand these diseases and to develop new treatment protocols based on the information provided by our MRIs so we can help patients as early as possible and improve their quality of life”. “Our idea is simple,” continued Edouard Duchesnay, NeuroSpin’s Director of Research in machine learning applied to neuroimaging: “in psychiatry, clinical interviews are currently used to establish a diagnosis and therapeutic strategy. We are looking to develop a kind of ‘thermometer’ capable of giving us an objective, a quantitative measurement of the problem, which should help physicians establish their diagnosis”. Being able to predict any evolution in their disease would be a significant advantage, and not only for patients,” said Edouard Duchesnay. “If we were to put money into tracking patients for several years, we would be able to determine who has the highest risk of becoming psychotic.”
“And this approach can also be applied to treatments”, added Josselin Houenou. “A typical example is lithium, which is administered to bipolar patients. A little less than half of all patients will not respond to this treatment, without us generally being able to identify why. We therefore progress by trial and error. It usually takes about one or two years before we can tell if the treatment is working or not, which means patients can lose all this time in the case where it doesn’t work.”
The image acquisition process lasts about an hour and involves either the 3T or 7T MRI, each located in a specific room with access strictly reserved for authorised staff and patients only. Chantal Ginisty is one of the radiography staff that carries out the MRI acquisition process. “In the case of patients with a psychiatric illness, there are a few small differences in the way in which we handle them compared with healthy volunteers. First, the psychiatrist is present during the entire examination to reassure the patient in the MRI machine. Next, we make sure that there are as few staff changes as possible for the patient. Last of all, these patients can perceive noise differently despite the protective hearing devices used, so we are particularly attentive to any stress they may feel.” She continued, “we are also careful about how we say things because some expressions can be seen as reassuring such as ‘I can see you with the camera’ or ‘I’m keeping an eye on you’ [in case there is a problem], while other sentences can be perceived as stressful to patients. Generally speaking, we review the patient’s psychological state with the physician before starting the MRI.” Other than these specific aspects, the MRI process follows the same sequence as that with healthy volunteers, making sure that they are as comfortable as possible in the MRI machine. “Remaining still during the entire scan is important for collecting exploitable data,” insisted Chantal Ginisty, “and comfortable patients are less likely to move during the process.”
Key figures
- Bipolar disorder, schizophrenia and autistic problems affect about 0.5% of the world’s population.
- About 15% of patients suffering from bipolar disorder will die from suicide.
- Less than 25% of patients suffering from schizophrenia work in an ‘open’ environment, i.e. conventional, unprotected environment.
Source:
https://www.sciencedirect.com/science/article/pii/S2215036621003953
Identifying bio-markers for diagnosis
After the MRI, the researchers have to analyse a large number of images in search of any abnormalities or engrams revealing a psychiatric illness. They are looking for the specific biomarkers of a pathology that can be compared against existing imaging data, as explained Cyril Poupon: “To identify these specific biomarkers in psychiatry, we rely on the identification of markers already catalogued in the neuroimaging database. There are four types of markers: anatomical, microstructural, functional and metabolic.” More specifically, he explained: “the markers collected during anatomical MRIs generally measure abnormalities in the shape of macroscopic anatomical structures like the cerebral cortex whose thickness or convolutions can be measured. Microstructural markers are collected during diffusion MRIs and provide information on the anatomical connectivity of the brain. These markers have various connections in the brain, which can be impaired in patients with psychiatric disorders. Diffusion MRIs also provide information on the microscopic organisation of brain structures. Other than abnormalities in the shape of structures, there can also be abnormalities in the composition of the cells forming these structures, which do not always result in changes to the shape of macroscopic structures. Functional markers are collected during functional MRIs that measure the activity of different regions in the brain. Brain activity in some areas can be heightened or diminished due to a psychiatric illness. Last of all, a host of biochemical reactions take place in the brain, which are only now being investigated thanks to ultra-high field MRI. For instance, it is now possible to measure the concentration of metabolites or electrolytes involved in neurotransmission.”
According to the specialist, “the future of neuroimaging medicine lies in collecting the maximum amount of information using these innovative, non-invasive procedures to identify and implement therapeutic strategies that are best suited to slowing down or effacing pathological traits revealing psychiatric disorders observable in neuroimages.”
Understanding how the treatments work
Another area of research at NeuroSpin focuses on treatment, more specifically on the lithium imaging method that is widely used to treat bipolar disorder. Though we use MRIs mainly for imaging protons (nuclei of hydrogen atoms) of the water making up almost 80% of our brain, we can equally detect and image less-concentrated chemical elements whether they are endogenous (naturally occurring in our body) or exogenous (having been administered orally or intravenously). Lithium has a stable isotope, i.e. lithium-7, which has specific magnetic properties that allow its distribution in the brain to be mapped, conditional to the development and use of specific antenna. MRIs coupled with lithium aim to help us better under the poorly known mechanisms of lithium salts in the brains of bipolar patients. According to Fawzi Boumezbeur, a specialist in heteronuclear spectroscopy and imaging, the relevance of this type of imaging is being able to determine “whether lithium really does penetrate the brain and at what concentration. We have seen that lithium does not spread homogeneously in the brain, which may indicate that there is an active mechanism causing this heterogeneity”. “We are also using lithium imaging to find out why this treatment does not work in some cases”, he continued. “We conducted a study on about thirty bipolar patients using the 7T MRI. Our objective was to understand the response of patients taking lithium, comparing those who responded positively against those showing fewer positive signs, in order to see whether the lithium concentration and distribution differed between the two cohorts. So far we haven’t been able to answer this question, but we intend to expand the study to cover a broader cohort of patients to improve our statistical credibility and test our assumptions”. “Unfortunately, another specificity of these psychiatric diseases is the extreme variability in the history of each individual and their symptoms. A cohort of only 30 patients will not cover the variability in such populations”.

Mental health © Pixabay
Anticipating risks using artificial intelligence
This is where Edouard Duchesnay comes into the picture. Relying on his expertise in data management and aggregation on hundreds of individuals, he and his computers are capable of generating snapshots of populations, drawing relevant information from the vast amounts of data by using artificial intelligence models. The principle involves analysing a massive quantity of imaging data from large cohorts, i.e. from a hundred to several thousand individuals, using algorithms to identify any biomarkers predicting the possible emergence of a disorder or indicating a suitable treatment protocol. “Neuroimaging reflects everything that has been going on in the brain; we are attempting to use artificial intelligence as a prognostic tool to monitor clinical signs. Therefore, genetic mutations, chronic stress or repeated exposure to stress, cannabis, or alcohol will produce very mild brain atrophy that is invisible to the eye, but are discernible in a conventional MRI. All these risk factors come together to build a global brain signature that AI can detect and predict - with a certain level of uncertainty - whether a pathology will emerge or not. Algorithms are currently able to predict transition to psychosis with an accuracy of 75-80% in young adults at risk, which is much higher than chance at 50%. This is therefore an important public health issue,” he pointed out. In more concrete terms, the principle is based on an image (corresponding to an individual) divided into voxels, i.e. the 3D equivalent of pixels used to described 2D. There are about 300,000 voxels in each image, to be multiplied by the number of individuals, which can range from several dozen to several thousand depending on the size of the cohort. The algorithms used in AI learn to predict the clinical evolution of patients based on the brain scans. These algorithms can also highlight different sub-groups in the clinical categories (schizophrenia, bipolar disorder, etc.) that each require a specific therapeutic strategy. “The real challenge will be to avoid overfitting, i.e. learning details that are correlated to what we want to predict but without these details actually reflecting a true causal link”, concluded Edouard Duchesnay.
If AI can predict the emergence of a pathology, then it can also predict the effectiveness of lithium treatments. This is the objective of the European R-Link project on which Edouard Duchesnay is working in collaboration with Fawzi Boumezbeur and Josselin Houenou. Initiated in 2021, this clinical trial sets out to monitor 300 bipolar patients who will all undergo an MRI, clinical assessment and genetic testing before any treatment protocol is prescribed (either because they have just been diagnosed, because they have taken lithium and stopped, or because they were taking another treatment). “After this, they will be given lithium before undergoing another MRI three months later, and being monitored for the next two years,” specified Edouard Duchesnay. “The question we want to answer is: can we find prognostic markers to predict the lithium response, either before treatment or during the first three months of treatment?”. The end goal is, of course, “to be able to predict whether lithium will work on someone or not”, stressed Josselin Houenou.
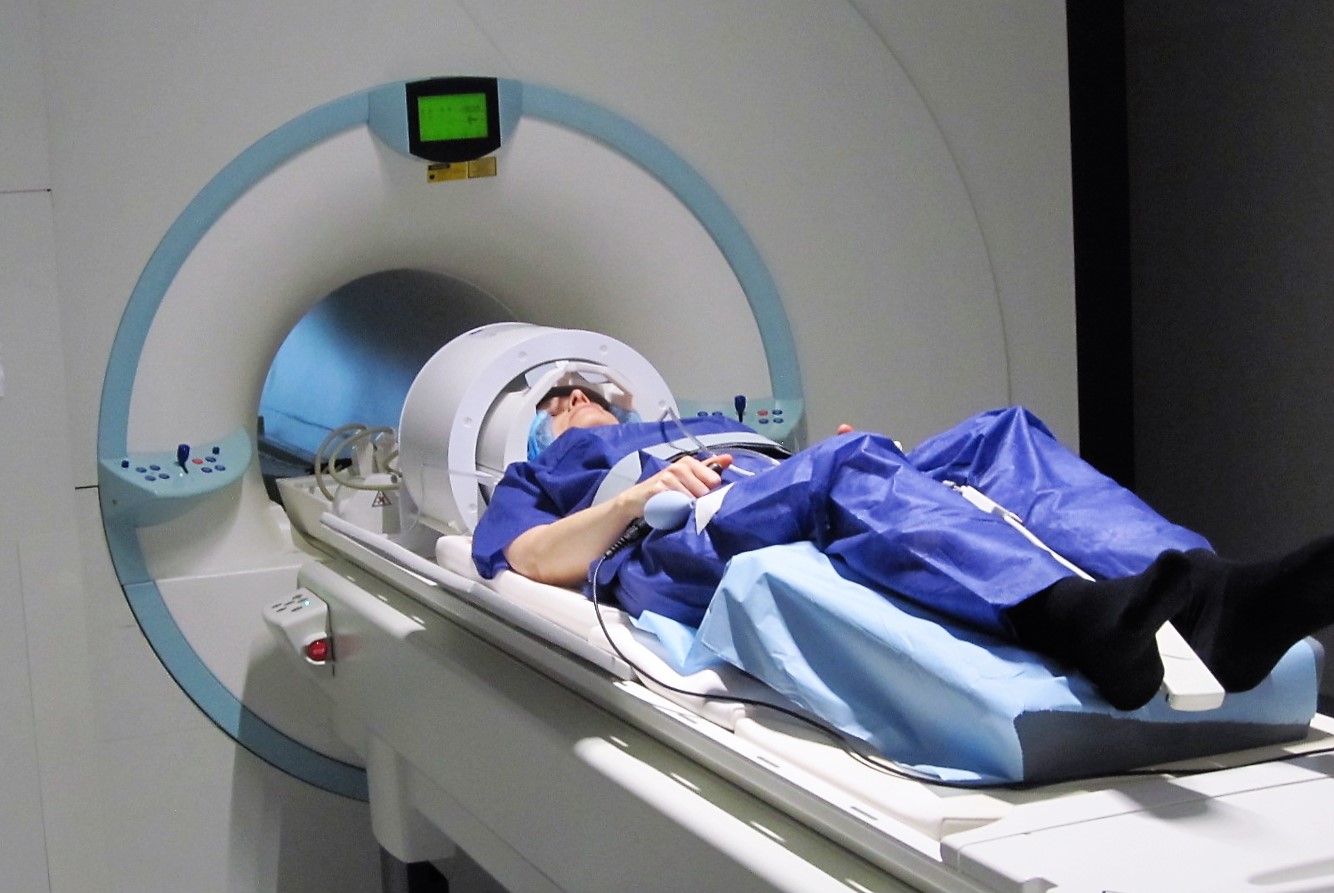
A volunteer in the 7T MRI at NeuroSpin. © C.Doublé / NeuroSpin / CEA
Developing new treatment protocols
On a much smaller scale, by identifying brain dysfunctions in patients with psychiatric diseases, MRIs can also lead to new treatment solutions. “NeuroSpin will be starting a clinical trial sometime in 2022 on bipolar disorders that will combine ‘neurofeedback’ with functional MIRs,” announced Josselin Houenou. “This method has enabled us to identify networks that do not function properly in these patients, especially when it comes to controlling emotions. These individuals will undergo a functional MRI using the 3T machine during which they will be shown emotionally charged images. We intend to observe the activity in the network of their brain that regulates emotions; we will then show them the same images again while simultaneously projecting the screen of their brain activity so they can try regulate their own emotions directly.” This veritable technical challenge was overcome thanks to the expertise of NeuroSpin’s methodologists in MRI sequence development. Neurofeedback (NFB) targets the symptoms of depression and anxiety that remain despite drug treatments, which can be very disabling and lead to relapse. “This is the first example of a treatment that has been developed thanks to neuroimaging”, stated Josselin Houenou. It is quite a demanding treatment because it requires four MRIs; if proven successful, this treatment will not be readily available to everyone. But we would like to be able to achieve the same effect by using a less-burdensome technique to MRI, such as electroencephalography (EEG) or magnetoencephalography (MEG)”. In the long term, the objective that involves combining all these approaches is to develop innovative, personalised medical treatments based on the analysis and use of large neuroimaging databases.
The different ultra-high field MRIs at NeuroSpin
The centre has six MRI machines: three preclinical MRIs providing 7, 11.7 and 17.2 Tesla (unit for measuring magnetic fields) used on small animals, and three clinical MRIs providing 3, 7 and 11.7 Tesla, currently being commissioned. These ultra-high magnetic fields provide a very high resolution, and the future 11.7T clinical MRI is targeting a resolution of about 100 microns. The 17.2T preclinical MRI is used to collect images with a resolution of 25 microns, i.e. the size of a neurone. In France, there are only three 7T MRIs and 200 3T MRIs out of about 850 clinical MRIs (most are 1.5T MRIs).