A team at the ‘Institut de Biologie Structurale’ (CEA/CNRS/UGA), in collaboration with the CIRI (Inserm/CNRS/ENS de Lyon/UCBL), discovered a novel interaction between two proteins from the measles virus. This ultra-weak interaction, involving only four amino acids situated in a very flexible and dynamic protein region, is essential for measles virus replication. This newly discovered interaction constitutes a new target to treat measles infection, but also infection by other viruses from the same family, that comprises highly dangerous human pathogens.
These results are published in Science Advances, on August 22nd, 2018..
The measles virus genome is covered by numerous copies of the ‘nucleoprotein’, forming a very long and protective helicoid structure. This envelope of the genome is essential for viral replication and is controlled by the ‘phosphoprotein’. This latter protein contains a surprising fraction of conformational disorder, meaning that it lacks a well-defined structure. The first 300 amino acids of the phosphoprotein are indeed disordered and highly flexible.
The function of these disordered domains remains elusive, in particular in view of the parsimonious use of genetic information by this family of viruses. Scientists at the IBS used high resolution nuclear magnetic resonance spectroscopy to show that this area of the amino acid chain of the phosphoprotein contains two nucleoprotein-interaction sites at the opposite ends of the disordered domain. These two interaction sites work together to maintain the nucleoprotein in a form that facilitates viral replication. In fact, mutation of only four amino acids in the newly discovered second interaction site, which interacts very weakly, inhibits measles virus replication.
“This interactions site constitutes a new target for treating measles virus infection or infection by other dangerous human viruses”, emphasizes Martin Blackledge, CEA research director and corresponding author (With Rob Ruigrok, UGA) of this study. In fact, this essential mechanism seems to be conserved among Paramyxoviruses, the family to which measles virus, but also mumps and Nipah viruses, belong. “All of these viruses contain this kind of disordered domains and our observations open up new perspectives for the development of drugs against this family of pathogens”, adds M. Blackledge.
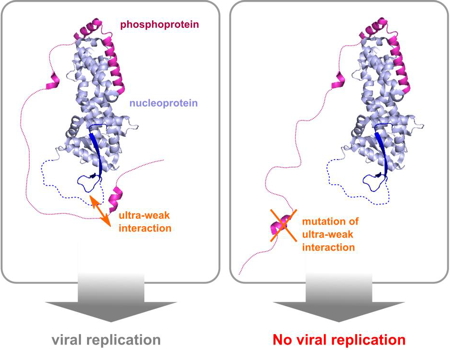
Scheme of the viral replication mechanism influenced by the expression or inhibition of the ultra- weak interaction.
© CEA
Measles virus
Measles virus belongs to the same family as Nipah virus, whose mortality rate for infected humans exceeds 70%. Even though the only reservoir of measles is the infected person, sometimes asymptomatic, and it is rapidly inactivated by ultra- violet light and various disinfectants, there is no specific antiviral treatment against measles. The disease is extremely contagious and still leads to about 100 000 deaths every year; in particular non-vaccinated young children can suffer from life threatening complications.
On the 20st of August, the World Health Organization (WHO) pointed out that more than 41 000 adults and children of the WHO European region were infected with measles during the first six months of 2018, which is more than twice the number of infections in the preceding year.

Measles virus nucleocapsid (top view).
© Gutsche I, Desfosses A, Effantin G, Ling WL, Haupt M, Ruigrok RWH, Sachse C, Schoehn G.