Understanding the mechanisms behind the resistance of pathogenic microorganisms is essential for developing new types of antibiotics. Researchers at CEA-Irig/LCBM/BioCat are analyzing the MsrPQ enzyme system, which could enable these bacteria to resist our immune system.
A few years ago, researchers discovered a new enzyme system called methionine sulfoxide reductase (MsrPQ) in certain pathogenic bacteria that enables them to resist hypochlorous acid (HOCl), which is produced by cells of the innate immune system such as macrophages and neutrophils.
Hypochlorous acid specifically causes oxidation of methionines in bacterial proteins, which leads to the loss of their structure and activity, and ultimately to the death of the pathogen. However, as a defense mechanism, these bacteria have developed the MsrPQ system, which repairs these oxidized methionines.
Using complementary approaches (directed mutagenesis, electron paramagnetic resonance spectroscopy, electrochemistry, high-performance liquid chromatography coupled with mass spectrometry, and AlphaFold), researchers at CEA-Irig/LCBM/BioCat were able to study the mechanism of MsrPQ in detail, particularly the central role of its membrane component MsrQ, which catalyzes a specific electron transfer reaction from the cytoplasmic space to the periplasm. In addition, they identified that MsrQ contains, in addition to heme* two new redox cofactors: a flavin* and a ubiquinone* (see Figure).
Beyond these fundamental aspects studied here, understanding how this new system works will enable us to better characterize bacterial virulence mechanisms and develop new types of antibiotics.
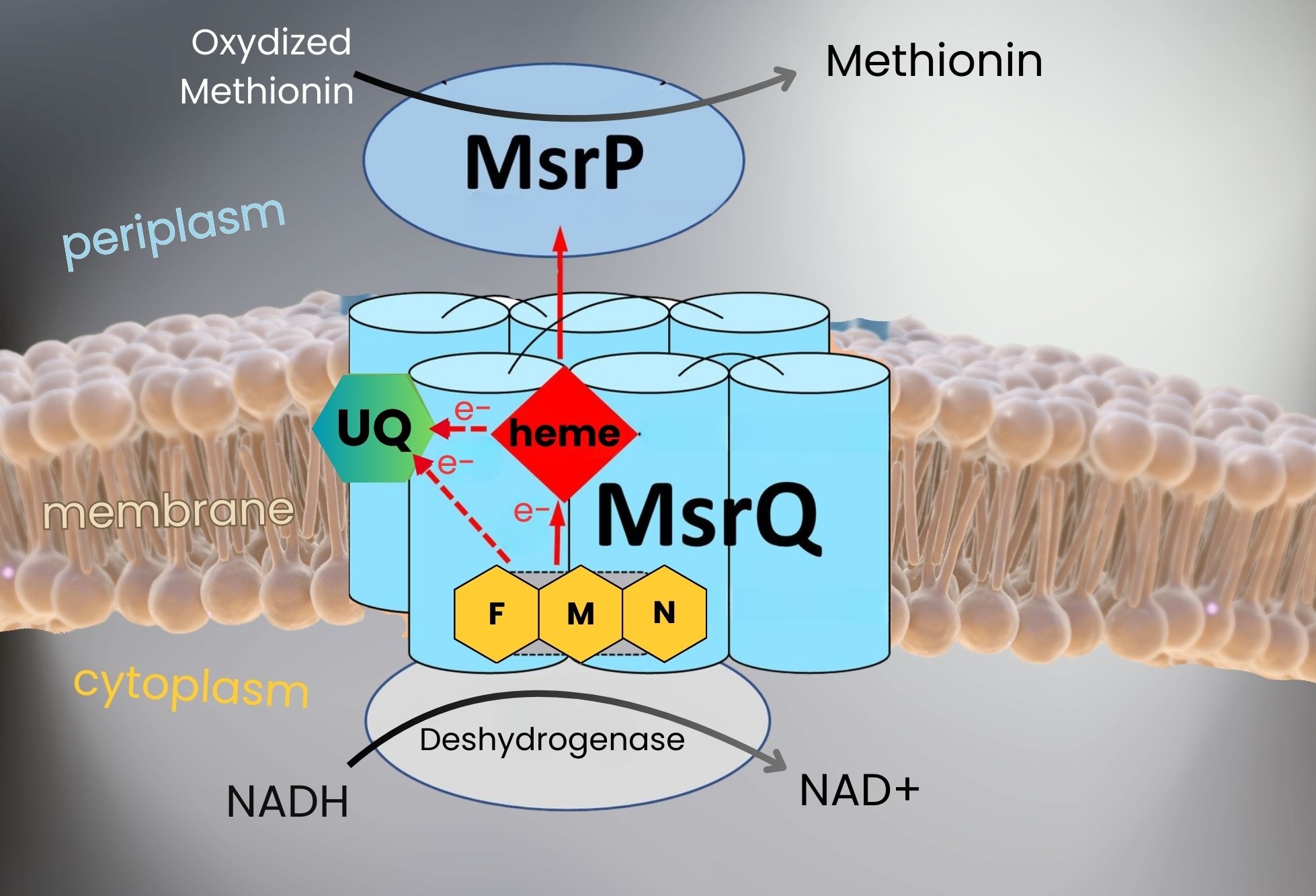
Figure: MsrPQ system repairs periplasmic methionines oxidized by HOCl. The membrane component MsrQ contains three redox cofactors (heme type b, flavin FMN, and ubiquinone UQ), two of which enable electrons to be specifically transported from the cytoplasm to the periplasm for the regeneration of oxidized methionines. © CEA
* Hems and flavins are biological redox cofactors which, when associated with enzymes, enable them to catalyze reactions involving electron transfers.
* Ubiquinone also fulfills this function, but is more commonly found in cell membranes.