Currently, less than one "drug candidate" in ten entering into clinical trials is commercialized in the end. This phenomenon leads to increasingly longer delays in reaching the market and higher development costs. However, the trend could be reversed through early identification of drug candidates that exhibit the best efficacy and minimal toxicity, by assessing their behavior in vivo (in animals but also in humans) from the earliest design phases. One technique consists in "labeling" these molecules by substituting certain atoms (H, C, F, etc.) with their isotopes (either stable or radioactive). This results in molecules that are labeled and thus detectable in the body. In this way, hydrogen (H), which is present in all organic molecules used in human health, may be replaced by deuterium (stable) or tritium (radioactive).
The rapid synthesis of labeled molecules, in a manner that is inexpensive and uses techniques that respect the environment, is a great challenge in isotope chemistry today. Indeed, introducing deuterium or tritium at a defined position of a molecule of interest generally requires the use of chemical precursors and a construction in several steps.
A team from the CEA-IBITECS, in collaboration with a laboratory combining INSA1, the CNRS and the Université Paul Sabatier (Toulouse), has developed a labeling method by "C-H activation". This method, feasible under mild conditions, is applicable to complex and fragile molecules and uses ruthenium nanoparticles. The researchers worked on the isotopic exchange of a C-H bond in the case where the carbon atom is chiral (i.e. when it carries four different groups). A chiral carbon is not superimposable on its mirror image (the same goes for our two hands). The vast majority of drugs and molecules of interest contain this type of carbon. The researchers succeeded in demonstrating the exceptional potential of ruthenium nanoparticles in order to achieve an isotopic exchange on a chiral carbon without modifying the initial three-dimensional structure of the molecule. In parallel with these results, the researchers conducted a computational chemistry study that helped reveal the entirely original reaction mechanism of this process. The latter opens new chemical and biological perspectives, and helps imagine new developments in labeling that could be applicable to basic research as well as the chemistry of drugs or materials.
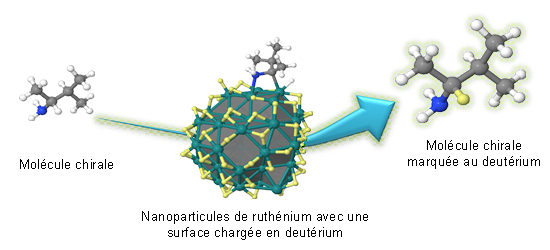
Isotopic exchange between the ruthenium nanoparticle (whose surface is impregnated with deuterium) and the chiral carbon, allowing the labeling of a molecule without modifying its initial three-dimensional structure. © Romuald Poteau (CNRS) and Grégory Pieters (CEA).
- Institut National des Sciences Appliquées of Toulouse