Copper is an essential trace element for humans, used for numerous physiological functions. Given the high toxicity of free copper, its transport and storage are very finely regulated in human cells. Over the last decade, copper has been shown to play an important role in tumorigenesis. Various anti-cancer therapies targeting copper are currently under development.
Researchers at CEA-IRIG, the Institute for Advanced Biosciences and the Free University of Brussels (ULB) have developed a new family of molecules that selectively bind and transport copper ions carrying a single positive charge (Cu⁺) which corresponds to the intracellular form of copper [1,2], across a biological membrane (Figures a-b). Some compounds in this family have shown anti-cancer activity in cultured cells. The most active compound, named Cuphoralix, was identified (Figure a) [2,3]. The expertise of CEA-IRIG researchers in
copper homeostasis* has helped to elucidate the mechanism of action of Cuphoralix in liver cancer cells. In collaboration with researchers at the ESRF synchrotron in Grenoble, elemental nano-imaging experiments showed that the Cuphoralix redistributes copper in cancer cells, resulting in copper poisoning, and ultimately cell death (Figure c-d).
The selectivity of molecules such as Cuphoralix developed for Cu+ transport has led to the discovery of a completely new anti-cancer mechanism of action. Considering that Cuphoralix has no specific intracellular target, cells cannot develop resistance mechanisms. This also makes it active against all types of cancer cells tested to date, including resistant and non-resistant cancer models, as well as those with or without an aggressive phenotype. All these properties make Cuphoralix a very promising molecule for anti-cancer therapy.
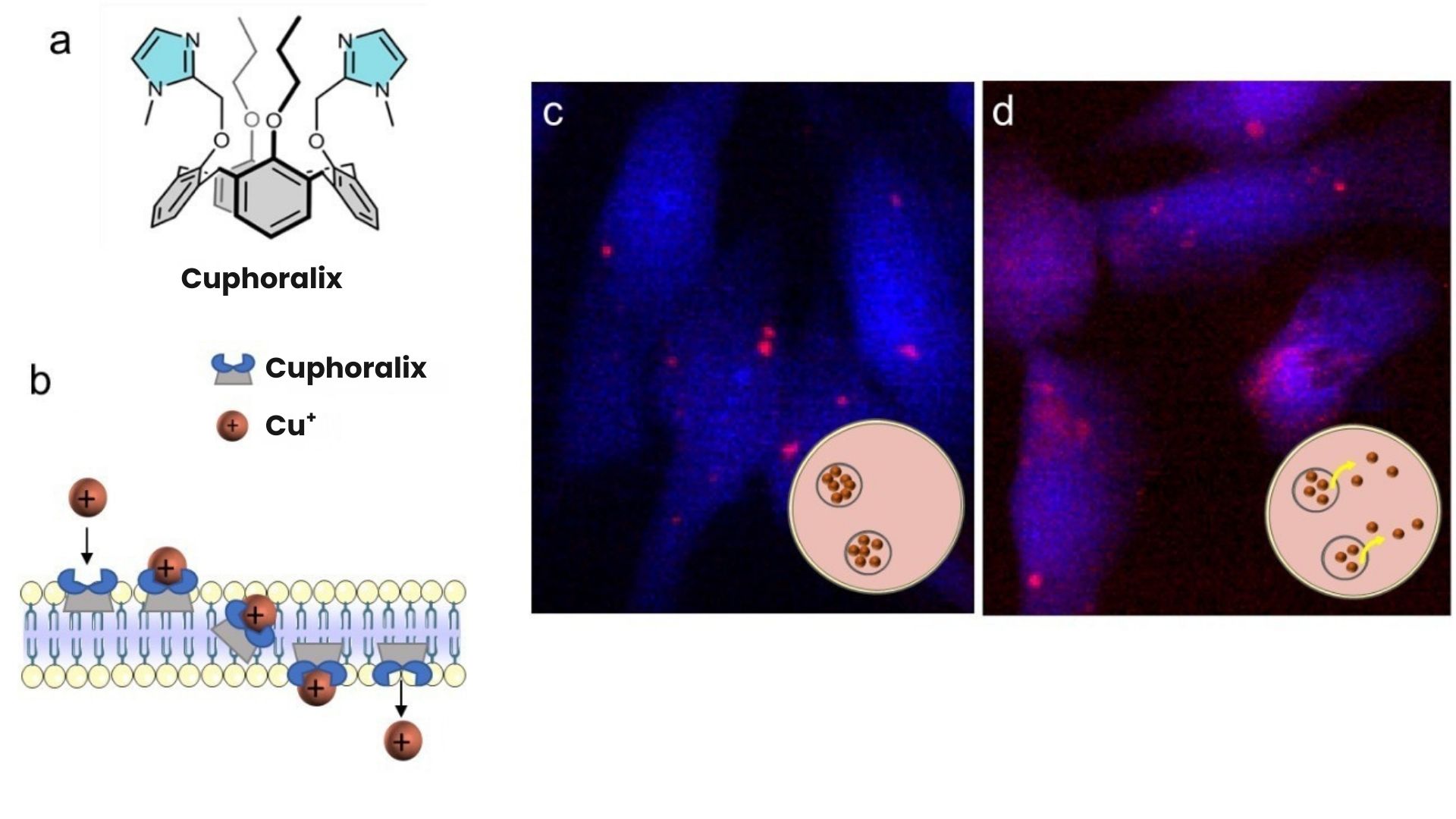
Figure: a) Structure of Cuphoralix.
b) Mechanism of Cu+ ion transport across a lipid bilayer by Cuphoralix.
c-d) X-ray fluorescence microscopy images showing the distribution of potassium (blue) and copper (red) in liver cancer cells exposed to 10 µM Cu (c) or 10 µM Cu and 100 nM Cuphoralix (d).
These maps show that copper is physiologically stored at high concentrations in intracellular vesicles (c, intense red spots) and that Cuphoralix prevents this storage, leading to a redistribution of copper throughout the cell with local concentrations 10 times lower (d). The insets show a schematic representation of the location of copper in cells.
* Copper homeostasis refers to the set of mechanisms that maintain the appropriate intracellular concentration and distribution of copper.