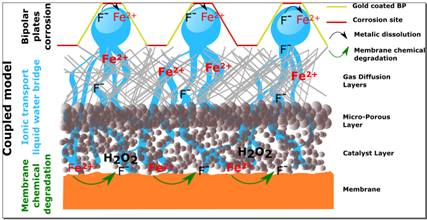
Generally speaking, degradation models allow researchers to look at single degradation mechanisms in isolation from each other. Unfortunately, this approach does not address potential interactions between the multiple mechanisms at work. Researchers at CEA-Liten, a CEA Tech institute, recently set out to test one interaction in particular.
Bipolar plate corrosion and membrane degradation are two of the best-known degradation mechanisms. The membrane is affected by a phenomenon called the Fenton reaction, in which metal ions act as a catalyst. The origin of these ions, however, is unknown. The researchers posited that plate corrosion could be the culprit. To test their hypothesis, they modeled the corrosion-induced emission of iron ions and the diffusion of these ions from the plates and through the cell to the membrane. Membrane degradation produces fluorides that stimulate plate corrosion, generating a coupling effect. This reverse fluoride transfer was also modeled. Finally, the researchers integrated the corrosion, membrane degradation, and ion diffusion models into the MePHYSTO fuel-cell simulator, part of the MUSES platform.
Modeling combinations of degradation mechanisms in this way is unique, and will now enable simulations of the interactions between the different phenomena at work in the stack as well as any snowball effects. Ultimately, the insights gained will help extend fuel cell lifespans and, therefore, reduce costs, by facilitating the selection of more resistant materials and by helping adapt operating conditions to limit degradation.
This research was part of a PhD dissertation by Imen Elferjani in partnership with MATEIS, an INSA Lyon lab specializing in corrosion.