Team presentation
The sulfur metabolism (fig 1) is highly conserved across species and occupies a position apart in the cellular function and homeostasis owing to the reactivity of thiols and the characteristics of the two sulfur-containing amino acids, methionine and cysteine. These amino acids, in addition to their roles in proteins, are precursor for the synthesis of two key cellular compounds: (i) S-adenosylmethionine (SAM), the major methyl donor for the methylation of nucleic acids, proteins and lipids; and (ii) glutathione (GSH), the most abundant antioxidant of the cell. The sulfur metabolism is tightly regulated to fit cell requirements as lack or over-accumulation of certain sulfur-containing compounds have dramatic impacts on cell physiology, organism's health and ageing.
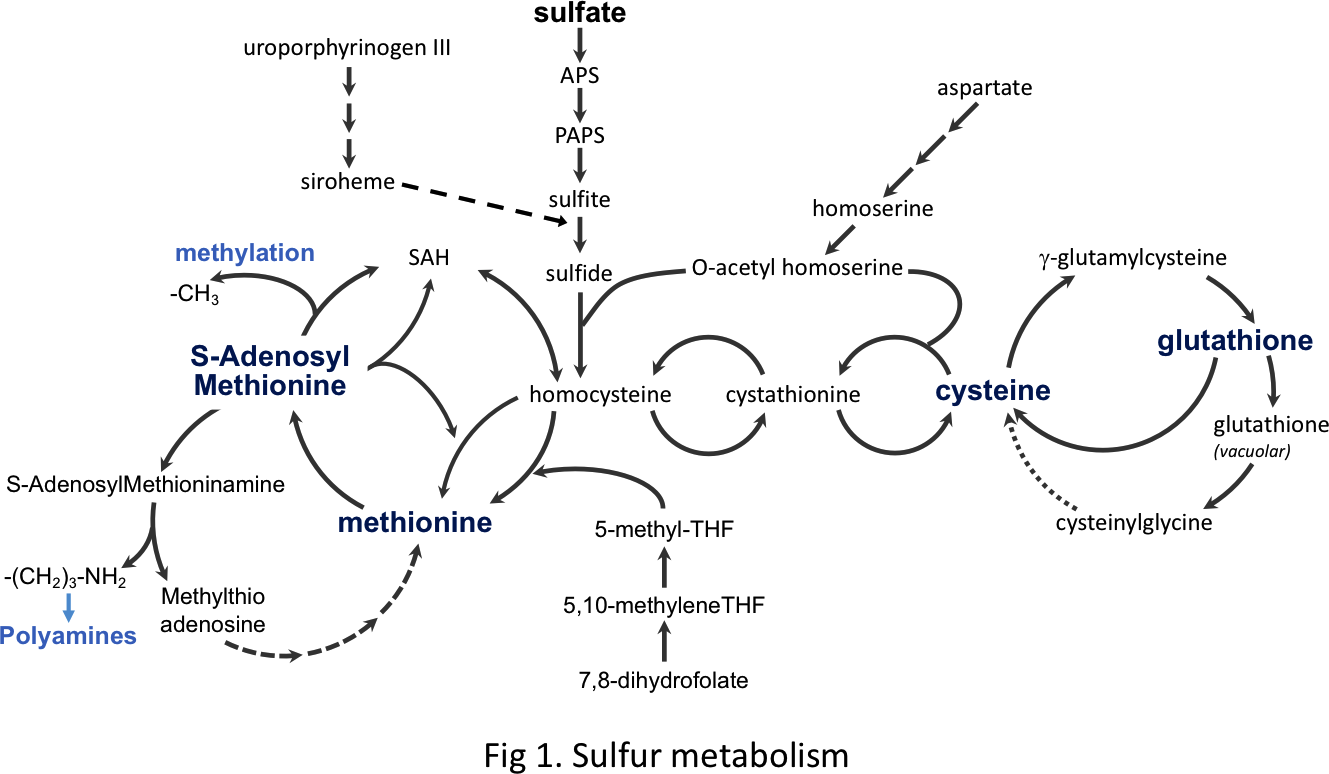
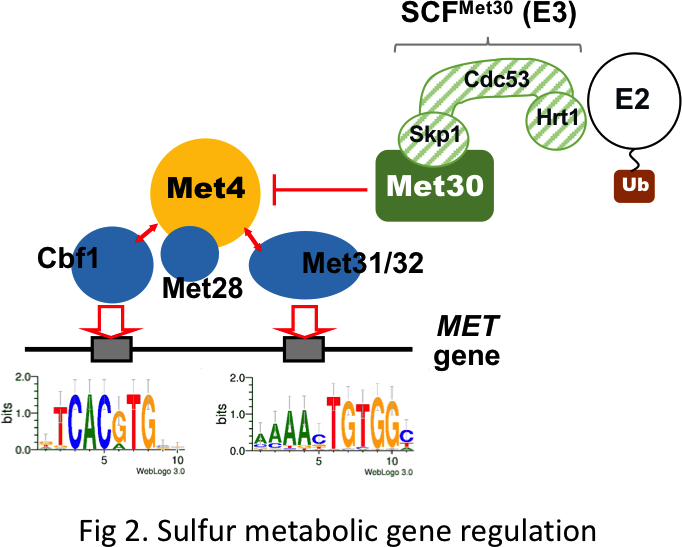
The sulfur-metabolism is a complex and integrated process controlled at numerous levels. For instance, we and others have shown in S. cerevisiae (i) a tight transcriptional control of the methionine and cysteine biosynthetic genes involving five transcription factors and an ubiquitin ligase (Kuras and Thomas, 1995; Kuras et al 1996, 1997, 2002; Cormier et al 2010) (fig 2), (ii) a specific sulfur-sparing program designed to replace some abundant enzymes with sulfur-depleted isoforms (Fauchon et al 2002; Pereira et al 2008), (iii) a dynamic control of the degradation of GSH which is also a sulfur storage compound (Baudouin-Cornu et al 2012). Altogether these regulations contribute to maintain the pool of essential sulfur-compounds at physiological levels regardless variations of sulfur sources and their availability in the cellular environment.
The main interest of our team is how cells respond and adapt to ensure survival over periods of starvation for sulfur nutrients. Indeed, when yeast cells are submitted to starvation for sulfur nutrients, they stop dividing but remain viable for weeks. This impressive capacity to survive correlates with a vast modification of gene expression leading to the induction or repression of several physiological processes. However, the precise mechanisms of adaptation, and whether they are complementary and coordinated, are still poorly understood. Our objective is to identify these mechanisms and to understand, at the cellular and molecular levels, how they contribute to cell survival during sulfur starvation. The capacity of yeast to resist sulfur starvation also raises the underlying question of the role of the sulfur compounds in the cell physiology and function.
We use targeted and global approaches to tackle these issues by addressing questions such as:
- What are the molecular actors required for survival and recovery under sulfur starvation?
- What is the role of autophagy in the process?
- Is there a redistribution/reallocation of the cellular pool of sulfur compounds during starvation?
- Is there an interplay between antioxidative sulfur compounds (GSH) and autophagy (knowing its role in elimination of damage molecules) in the protection against oxidative stress injuries and ageing diseases?
These studies will help to better understand the impact of severe nutritional (sulfur) stress on the cell behaviour and function, as well as their consequences on cellular health. They could also contribute to the identification of new targets for anti- or pro-oxidant therapy.
Key words: Sulfur, methionine, S-adenosylmethionine, Glutathione, metabolism, nutritional control, oxidative stress, redox homeostasis, adaptation, autophagy, transcription regulation, yeast