REsearch topics
CROSSING OF THE HUMAN BLOOD-BRAIN BARRIER
The blood-brain barrier (BBB), located at the level of the endothelial cells of the cerebral capillaries, has a restricted and selective permeability due to the presence of tight junctions in the inter-endothelial spaces. It thus controls the composition of the cerebral interstitial medium. This relative impermeability poses a major problem for the passage of many drugs into the central nervous system. The important pharmacological issue of BBB crossing has led us to the experimental modeling of this barrier using induced pluripotent stem cells. We hope to better understand its role in rare and neurodegenerative brain diseases and to study the kinetic parameters of the passage of drugs destined for the nervous system upstream of the drug discovery process.

Blood-brain barrier : blood-brain interface © Ben Brahim Mohammed/CEA
Associated projects
Modeling the human blood-brain barrier for screening and selection of therapeutic molecules : i) collaborative project with Servier Laboratories (multi-year collaboration); ii) MICROCOSM Project : a microfluidic system for drug candidate research and discovery.
BRAIN ORGANOIDS IN NORMAL AND PATHOLOGICAL CONDITIONS
We develop vascularized and/or on-chip brain organoids in a physiological or pathological context (rare brain diseases, primary or secondary tumors) for: (i) the search for biomarkers of pharmacological efficacy for personalized medicine; (ii) the study of neuronal and glial cell interactions in brain pathologies; (iii) the pharmacological evaluation of drug candidates.
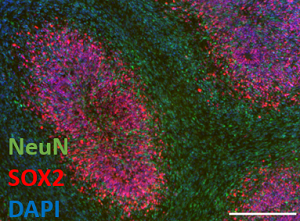
Associated project
Modeling of brain organoids for the validation of molecular signatures in rare neurological diseases : associated project with the American Association for Congenital Creatine Deficiency (ACD)
INNOVATIVE STRATEGIES FOR DRUG DELIVERY TO THE BRAIN
Implementation of innovative strategies for the delivery of drug candidates and diagnostic molecules, small molecules and biotherapeutics, in the brain.
 Delivery strategy for drug candidates in the brain © A.Mabondzo/CEA
Delivery strategy for drug candidates in the brain © A.Mabondzo/CEA
TRANSLATIONAL RESEARCH IN NEUROSCIENCE
Accelerate the transfer of innovation from research to the clinic through technology transfer, the development of industrial partnerships or the creation of spin-offs for the following pathologies : hypoxic-ischemic encephalopathy, glioblastoma, melanoma, breast cancer with brain metastases, creatine transporter deficiency and cerebellar ataxia.
Associated projects
- NIH project (Co-principal investigator) (2021-2026) : Purine derivatives and hypoxic-ischemic encephalopathy: characterization of neuroprotective activity in a hypoxic-ischemic model. This project is implemented in a consortium involving the Brown University and the University of Washington (USA).
- ANR-Dual Mab project (Partner of the consortium Dual Mab) (2020-2023) Dual fluorescent and isotopic imaging of glioblastoma stem cells.
- ANR-INSPIE project (Principal investigator) (2021-2024) : Validation of the molecular mechanisms of purine derivatives using human brain organoids. Project in partnership with the Robert Debré Hospital and the University of Gothenburg in Sweden.
- CRENATHER project (Principal investigator) (2018-2021) This project, funded by the Fondation Lejeune and the Fondation Maladies Rares, aims to identify protein signatures in different brain areas related to the improvement of cognitive abilities in mice with creatine transporter deficiency and treated with our drug candidate: creatine dodecyl ester.
- Brain-Iway project (Principal Investigator) (2021-2022) : This project, funded by Idex Paris-Saclay, aims to deliver Trastuzumab to the brain via the nasal route for the treatment of brain metastases in breast cancer.
- INCA project (Partner of the PAIR consortium) (2022-2026) Preclinical evaluation of the delivery of an internal vectorized radiotherapy targeting endothelin receptor-expressing glioblastomas to the central nervous system.
- ANR-Glyco project (Partner) (2021-2024) tritium-NMR - Metabolic activity, coupling neurons/glial cells (SCBM/SPI/Neurospin)
- Collaborative project with CERES BRAIN THERAPEUTICS (2019-2024) Preclinical development of CBT101 for creatine transporter deficiency.
- NIH project (Co-principal investigator, submitted) in partnership with the University of Cincinnati (Ohio) : Brain organoids and creatine transporter deficiency: Validation of molecular signatures.
- NIH project (Co-principal investigator, submitted) : Optimization of the pharmacokinetic properties and pharmacodynamics of purine derivatives for the treatment of hypoxic-ischemic encephalopathy.
FINANCIAL RESOURCES
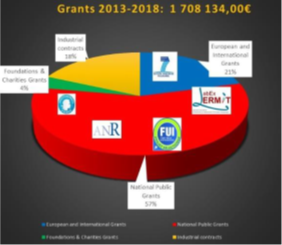 Between 2013 and 2018, the laboratory generated 1.7 M€. From 2019 to June 2021, we have obtained 1.8 M€ of revenues from international NIH, national ANR, INCA, industrial contracts and calls for projects from the University of Paris-Saclay.
Between 2013 and 2018, the laboratory generated 1.7 M€. From 2019 to June 2021, we have obtained 1.8 M€ of revenues from international NIH, national ANR, INCA, industrial contracts and calls for projects from the University of Paris-Saclay.
Financial resources of the team from 2013 to 2018 © A.Mabondzo/CEA
Equipment
The LENIT disposes of :
|  © F.Rhodes/CEA
|  © F.Rhodes/CEA
|
- Biosafety Level 2 laboratories for eukaryotic
cell culture.
- A platform of in vitro blood-brain barrier models (human and rodent)
- A level 2 biosafety laboratory for prokaryotic cell culture
- Equipment to perform cellular and molecular analyses (flow cytometry, icyler for quantitative PCR)
- Equipment for cell imaging, histological studies (Vibratome, cryostat...)
- Equipment for animal pharmacology studies (chamber for hypoxia-ischemia, device for stereotaxis)
- Equipment for the purification and characterization of proteins (spectrofluorometer, Dynamic Light Scattering, study of the stability of proteins by thermal denaturation, medium pressure chromatography...)
PARTNErS
- CEA : NeuroSpin (Drs Fawzi Boumezbeur, Sébastien Mériaux, Louisa Ciobanu), SHFJ (Drs Nicolas Tournier, Charles Truillet), SCBM (Drs Sophie Le Feuillastre, Frédéric Taran), Li2D (Dr Jean Armengaud), MIRCen (Dr Philippe Hantraye), SEPIA (Drs Jean Philippe Deslys, Franck Yates).
- Collège de France (Dr Elena Dossi).
- INSERM (Pr Pierre Gressens)
- USA a. Brown University (Pr Barbara Stonestreet); b. Cincinnati University (Pr Matthew Skelon); c. Washington University (Pr William Banks); d. Women and infants Hospital, Providence (Pr Barbara Stonestreet)
- Fraunhover University (Dr Yulia Kiian)
- Clinical Sciences Department, College of Medicine, University of Sharjah, United Arab Emirates (Dr Rifat Hamoudi)
- Division of Surgery and Interventional Science, University College London, W1W 7EJ London, United Kingdom (Dr Rifat Hamoudi)
- Department of Pharmacy Practice and Pharmacotherapeutics, College of Pharmacy, University of Sharjah, United Arab Emirates (Dr Rania Harati)
- Servier laboratories, INSERM Paris-Diderot
- Institute of Molecular Chemistry-University of Burgundy,
- Paris-Descartes university (Pr Hervé Galons)
- Liège university
- Necker hospital(Pr Pascale de Lonlay)
- Lyon civil hospice (Pr Vincent Desportes, Dr Aurore Curie)
- Institut du Cerveau et de la Moelle (ICM), Pitié-Salpêtrière hospital (Dr Stéphane Hunot)
- Robert Debré hospital (Pr Odile Boespflug-Tanguy)
LENIT SPIN-OFFS & Partners