OBJECTIVE 2: Position HLA-G as a diagnostic marker of graft stability and a therapeutic tool for preventing rejection in allogeneic therapies.
Since SRHI first described HLA-G expression in the context of heart transplantation in 2000, our team has demonstrated that after solid organ transplantation (heart, lung, kidney, and liver), HLA-G expression was significantly observed in stable patients and not in patients experiencing rejection. In line with this observation, the research team found a decrease in anti-HLA antibodies, known to be involved in allograft rejection, in HLA-G+ recipients. In this context, HLA-G is present in transplanted cells (immunohistochemical analysis of the graft) and in plasma (where soluble forms of HLA-G are measured by ELISA). These studies have led to HLA-G being considered as a marker for identifying patients at low risk of rejection and as a therapeutic target in the development of new anti-rejection treatments.
Role of the HLA-G molecule in lung transplantation as a predictive marker of graft survival and therapeutic target for anti-rejection treatment
In recent years, the SRHI team has focused its efforts on understanding the diagnostic and therapeutic potential of HLA-G in lung transplantation (LTx) and continues to do so. After LTx, the prognosis is associated with the onset of chronic rejection in the form of bronchiolitis obliterans syndrome (BOS). It is therefore important to have markers that predict graft progression. In this regard, the SRHI had previously shown that HLA-G expression by the grafted tissue is associated with allograft acceptance. With a view to identifying non-invasive markers, we recently described (i) an increased proportion of peripheral CD4+CD57+ILT2+ T lymphocytes during the first year following LTx (Brugière, JHLT 2022 and CEA-FOCH patent), and (ii) increased plasma levels of vesicular HLA-G (Brugière, Transplantation 2025 and CEA-FOCH patent) can distinguish patients who will experience subsequent rejection from those who will remain stable 3 years after transplantation. The future project aims to explore the nature, origin, function, and clinical use of these extracellular vesicles harboring HLA-G (HLA-G+ EVs), based on the hypothesis that they originate from the graft, are tolerogenic, and could serve as a non-invasive predictive marker and a new therapeutic tool against rejection. This project is being conducted in collaboration with (i) Foch Hospital (Olivier Brugière, LTx department, access to the national multicenter COLT cohort) and (ii) the Particles and Complex Systems Laboratory at the Faculty of Saints-Pères (Florence Gazeau), an expert in biology and the therapeutic use of EVs. (i) A flow cytometry analysis will be performed to define the origin of EVs from LTx patients, with specific markers for epithelial and immune cells, and (ii) immunological tests (MLR, cytokine secretion, etc.) to evaluate the function of HLA-G+ EVs from HLA-G+ cell lines (proof of concept) and LTx patients (clinical relevance).
HLA-G and allogeneic cell therapy
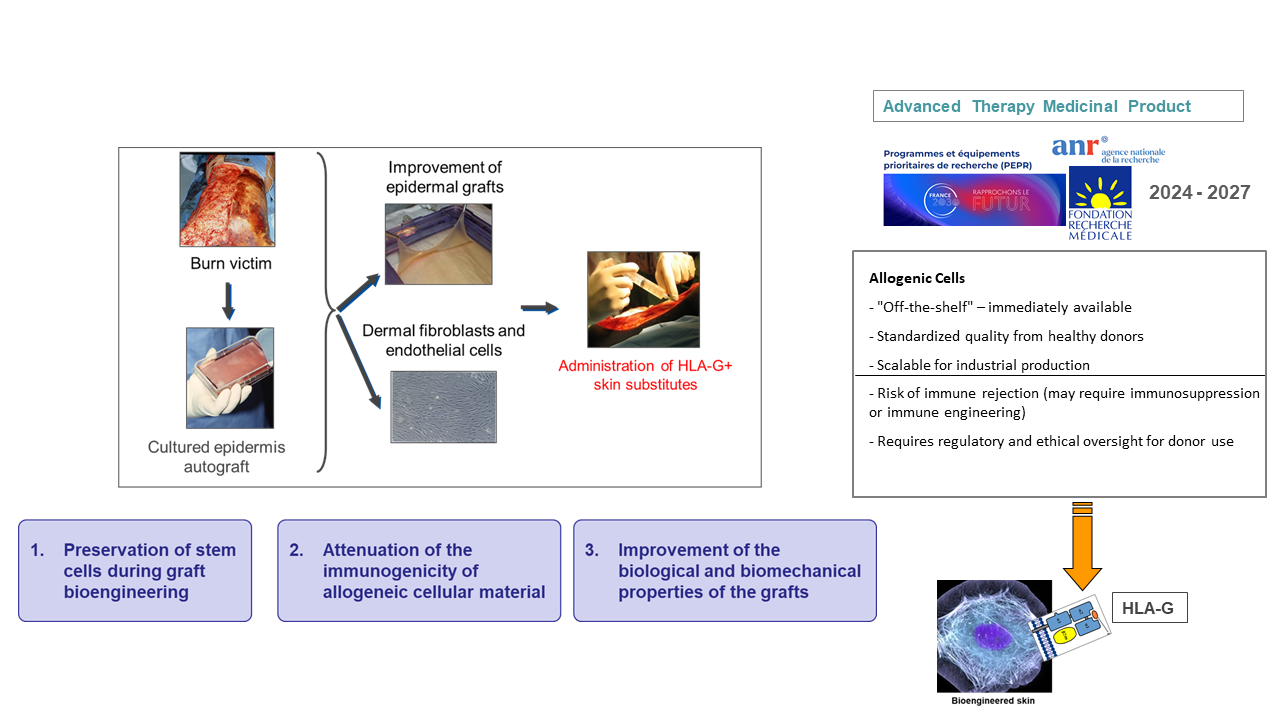
This project builds on our recent findings on the role of HLA-G in enabling keratinocytes in primary culture to acquire immunomodulatory properties (Mestrallet, Cells 2021 and Front Immunol 2022). We are currently studying HLA-G-mediated immunomodulation to improve the tolerance of autologous skin grafts, which are generally performed in an inflammatory context, or allogeneic skin bio-bandages, which face immune rejection.
Skin grafts are used in various surgical specialties, such as orthopedics and plastic and maxillofacial surgery. They play a central role in the treatment of severe burns. However, innovations in tissue engineering are essential to enable standardized bioproduction of skin grafts on a larger scale, ensuring total safety for patients. This project focuses on one of the factors determining their therapeutic efficacy: keeping under control the immunological parameters that could compromise long-term engraftment, which currently limits the use of allogeneic bio-dressings. This project is part of a strategy aimed at designing a new generation of bioengineered composite skin with greater immune tolerance than current graft models.
This approach aims to exploit the tolerogenic properties of HLA-G, currently being studied in human epidermal keratinocytes. As part of this project, we propose to implement this concept in a three-dimensional bioengineered skin graft comprising the two compartments of skin tissue, the dermis and the epidermis. The immunomodulatory action mediated by the membrane isoform of HLA-G (i.e., HLA-G1) should promote immunotolerance to the grafted epidermis. HLA-G-expressing fibroblasts will also be used to colonize the dermal component of the organoids and test their potential as vectors of an immunomodulatory signal in the deep part of the tissue. One of the advantages of this approach is the targeted impact of HLA-G. This effector does not alter the defense against pathogens, the expected effect being the protection of transplanted cells. The prospects concern the improvement of skin grafts in severely burned or irradiated patients, as well as regenerative and reconstructive surgery. This project is funded by the FRM (2023-2027) and is led by Nicolas Fortunel (CEA-Evry-LGRK), an expert in skin biology. It brings together teams of experts in HLA-G immunology (our team), preclinical (porcine) and clinical transplant studies (Alexandre Lellouch, Tenon Hospital, Paris), and skin replacement (JJ Lataillade and Stéphane Banzet, IRBA/CTSA, Percy Hospital). Capitalizing on the work accomplished and experience gained during this first project on tissue engineering with HLA-G, we hope to transpose this approach in the future to obtain solid, HLA-G-expressing, tolerogenic organs.