Development processes of living organisms have been studied at three levels: tissues (morphogenesis), cells (proliferation and differentiation) and molecular components, as found at precise concentrations within defined spatiotemporal conditions (morphogen gradients) for these latter. Morphogens have proved to be important at all three levels, as they were quickly shown to control both cellular specialization and tissular organization and thus offer a mechanistic view for events controlling morphogenesis.
Retinoic acid (RA), a vitamin A metabolite, is a key morphogen in vertebrate embryogenesis, particularly as concerns the development of the nervous system, as it induces neuronal differentiation of stem cells. At the molecular level, this process involves the binding of the metabolite to aptly-named retinoic acid receptors (RARs), which play a major role in gene expression.
Three genes (RARA, RARB, RARG) code for the receptors in mammals, thus providing the cell with a range of receptor subtypes (RARα, RARβ et RARγ) able to influence the cell's destiny. Currently however, the gene regulation programs falling under the control of these various receptors have not been sufficiently identified.
Researchers have nonetheless successfully created synthetic ligands with the ability to specifically activate each of these receptors. Thus, in an earlier work, researchers from the Genomics Metabolics 8030 mixed research unit (Genoscope) were able to show that BMS753, a RARα-selective ligand, induced neuronal differentiation, but BMS641 and BMS961, the comparable RARβ- and RARγ-selective ligands, did not1.
However, in a new work published in Life Science Alliance, that team has went on to show that the RARβ- and RARγ-selective ligands,when deployed together, are able to restore neuronal differentiation in murine stem cells, and notably via the reactivation of gene regulation programs normally controlled by the RARα receptor.
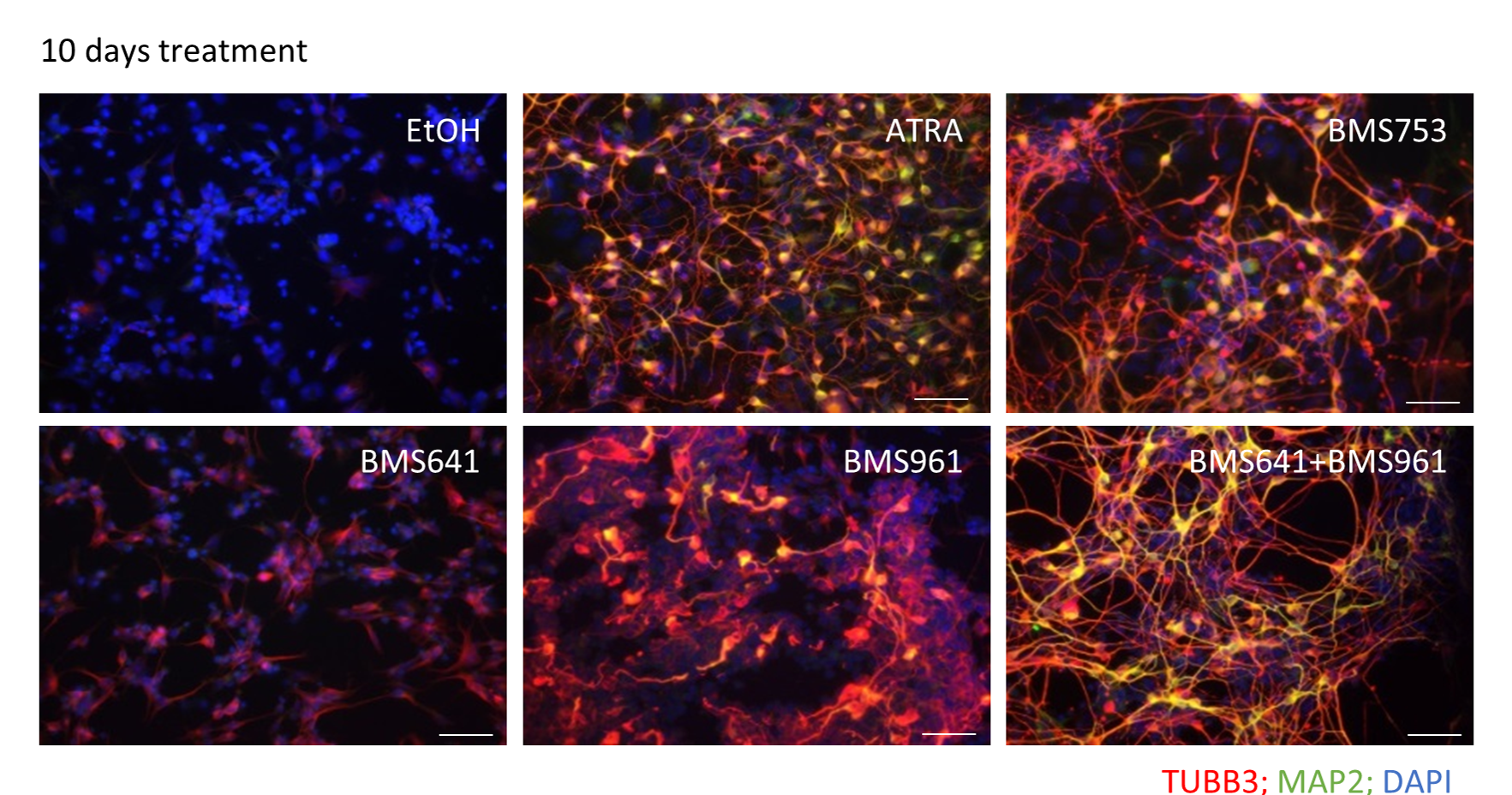
Beyond the neuronal differentiation process, the researchers also explored cell specialization, reporting the appearance of glial cells and a range of nerve-cell subtypes, including oligodendrocyte precursors, resulting from the murine stem-cell model.
By marrying cell engineering and transcriptomics, the Genoscope team established a more precise method for exploring the gene regulation programs under the control of the RAR receptors and particularly the role of these latter in neuronal differentiation.
With their efforts to build an englobing molecular vision of the RAR-controlled gene regulation programs involved in neuronal differentiation and more largely in brain tissue morphogenesis, the researchers aim to better understand the roles of these processes in adult cerebral homeostasis, and, by extension, their roles in certain neurodegenerative diseases.