Non-small cell lung cancer (NSCLC), the leading cause of cancer death worldwide, has a high degree of tumor heterogeneity, which is a source of treatment resistance. Imaging is essential for diagnosis and stratification of patients but has limitations. Advanced methods for extracting parameters from standard clinical images (radiomics) are currently being evaluated, but are limited by the fact that their structural and functional information is intrinsically very limited. In this context, quantitative multiparametric PET/MRI imaging offers unique possibilities for tissue and functional characterization of tumors at the intra-tumoral regional level.
Taking advantage of the unique multiparametric capabilities of the SHFJ PET/MRI device, a simultaneous dynamic multiparametric PET/MRI imaging protocol was developed and prospectively applied to 11 patients with histologically proven NSCLC. For each tumor, 12 tumoral parametric maps were generated, including full kinetic modeling of carbohydrate metabolism (PET), cellularity via apparent diffusion coefficient (diffusion MRI), tissue magnetic properties (T1/T2 MRI relaxometry), and full kinetic modeling of tumoral vascularization (perfusion MRI). Statistical partitioning (Gaussian mixture model) was applied on the data set to identify groups of voxel-supervoxels with similar PET and MRI multidimensional characteristics. A supervised machine learning algorithm (eXtreme Gradient Boosting, XGBoost) was finally trained and validated on the data (multidimensional features as inputs and corresponding supervoxels as outputs, 10 cross-validations) to identify the dominant PET/MRI multidimensional features of these supervoxels at the group and individual tumor level.
Statistical partitioning applied to the data set (17 316 voxels) revealed 3 main groups of voxels sharing the same multidimensional characteristics defined by the 12 PET/MRI structural and functional parameters. Of the 12 PET/MRI parameters, 4 clinically relevant parameters (metabolism and perfusion/vascularization) predicted the overall supervoxel characteristics with 97% accuracy.
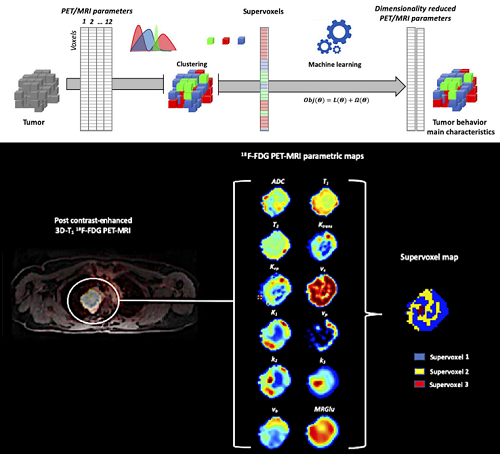 Top diagram: for all tumors, multiparametric PET/MRI data are statistically partitioned (Gaussian mixture model) in order to identify groups of voxels with the same multidimensional characteristics (supervoxels). A dimensional reduction by supervised machine learning allows to identify the most relevant multimodal parameters characterizing these supervoxels.
Top diagram: for all tumors, multiparametric PET/MRI data are statistically partitioned (Gaussian mixture model) in order to identify groups of voxels with the same multidimensional characteristics (supervoxels). A dimensional reduction by supervised machine learning allows to identify the most relevant multimodal parameters characterizing these supervoxels.
Bottom image: from a simultaneous dynamic multiparametric PET/MRI acquisition of a tumor, 12 maps are generated. These maps reflect different regional structural and functional characteristics of the tumor. Statistical partitioning allows to structure the tumor into 3 meta-regions / supervoxels, defined by the multidimensional characteristics of the tumor. (© Florent Besson, BioMaps/SHFJ/CEA)
In conclusion, the simultaneous dynamic multiparametric PET/MRI quantitative approach is clinically feasible to characterize the complex biological behavior of tumors. This proof of concept applied to NSCLC should open new perspectives in image-based precision medicine.
Contact : Florent Besson (florent.besson@aphp.fr)
Affiliations of key study contributors :
- Université Paris-Saclay, CEA, CNRS, Inserm, BioMaps, SHFJ, Orsay ;
- École de médecine, AP-HP, CHU Bicêtre, Le Kremlin-Bicêtre ;
- GE Healthcare ;
- Laboratoire de Mathématiques d'Orsay, CNRS, Université Paris-Saclay, Orsay ;
- Inserm UMRS999, Hôpital Marie Lannelongue, Le Plessis-Robinson ; Hôpitaux Universitaires Paris-Saclay, AP-HP ;
- Institut d'Oncologie Thoracique, Gustave Roussy, Université Paris Saclay, Villejuif