Isotopic labeling
Development of new methodologies for the labeling of biomolecules using Hydrogen Isotope Exchange
Deuterium/tritium labeling of biomolecules starting from synthetic precursors is usually time and atom-consuming, expensive and sometimes erratic. Biology but also therapeutic innovation both need the development of more robust, versatile and reliable methods for isotopic labeling that can use the native molecule without preliminary modification. In collaboration notably with Dr. B. Chaudret and Pr. R. Poteau at INSA Toulouse (CNRS/Univ. Paul Sabatier), we started to work on the study of ruthenium nanoparticles-catalyzed H/D and H/T exchanges. In particular, we demonstrated the unprecedented potential of these nanoparticles to catalyze the H/D isotopic exchange on a series of nitrogen containing compounds (aliphatic and heterocyclic) and thioethers. Reactions were performed in very mild conditions (1-2 bar, 20-55°C) and in various solvents (either organic or aqueous). This strategy was shown to be fully applicable for the stereoretentive deuterium labeling of drugs, amino acids and peptides and was applied to complex structures such as pharmaceuticals or oligonucleotides. Moreover, our group also maintains an expertise in terms of tritium labeling of biomolecules through more classical pathways, in particular in the framework of collaborative research activities with academic or industrial partners (Roche, Sanofi, Merck).

Substrate scope for C-H deuteration / tritiation © G. Pieters/CEA
Collaborations:
- Academic: Pr. P. Couvreur (Université Paris-Sud) / Dr. B. Chaudret (INSA Toulouse) / Pr. Romuald Poteau (INSA Toulouse) / Dr. P. Lesot (Université Paris-Sud)
- Industrial: Sanofi, Roche, Merck
Development of a new strategy using tritium NMR for the 3D structure resolution of small molecules interacting inside huge macromolecular complexes
Atomic scale resolution of the 3D structure of small molecules interacting inside a huge molecular complex (self-assembly, membrane peptide, non-crystallized ligand-macromolecule complex) is a prerequisite to understanding their chemical and/or biological activity. Presently, there is no efficient method to solve the atomic structure of such non-crystalline complexes. In collaboration with Drs. M. Paternostre (CNRS) and T. Charpentier (CEA), we initiated the development of a new strategy which combines tritium labeling, solid-state tritium NMR (spin ½) and molecular modelling in order to resolve the structure of non-crystalline molecular complexes and assemblies. The proof of concept was done on small rigid molecules and showed it is possible to measure interatomic distances up to 14 Å with an unprecedented precision. Successive determinations of intramolecular inter-tritium distances on crystalline Phe-Phe, the structure of which has been resolved by X-ray diffraction, is now under progress and will be then applied to the mysterious Phe-Phe nanotubes. This new strategy of 'crystallography by NMR' will be also used to solve the atomic structure of other biologically relevant nano-objects such as antimicrobial peptides, anti-amyloid compounds.
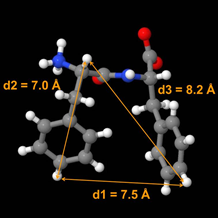
Atomic-scale 3D structure resolution though successive determinations of inter-tritium distances
© SCBM/CEA
Collaborations:
- CEA: Dr. T. Charpentier, Dr. M. Paternostre, Dr. Y Boulard
Characterization and study of materials using tritium
Nanodiamonds are nanosized carbon frameworks which display particular structures and properties. Recent studies suggest that after hydrogenation, nanodiamonds can be used as molecular scaffolds for the design of nanocatalysts capable of performing particular chemical reaction. They can also be used as nanodelivery systems carrying drugs and other bioactive molecules. Moreover, the fluorescent properties of certain nanodiamonds suggest that they could be used as molecular probes for diagnostic imaging. In cooperation with Drs. T. Charpentier and J-C. Arnault (CEA), we have investigated the surface chemistry of nanodiamonds reduced by a tritium plasma or using thermal annealing in order to determine, by tritium solid-state NMR, the position and number of hydrogenated motifs usable for engineering the nanodiamond surface.

Microwawe-generated tritium plasma for the tritium
labeling of nanodiamonds © SCBM/CEA
Collaborations:
- CEA: Dr. J.C. Arnault, Dr. H. Girard, Dr. M. Mermoux, Dr. M. Schlegel
Tritium chemistry for applied research with high societal impacts
Initiation of plasma experiments (tentatively in 2025) in the international experimental thermonuclear fusion reactor ITER settled in Cadarache should allow to make the transition of experimental plasma physics to full-scale electricity-producing fusion power plants. However plasma ignition inside the tokomak necessitates supplementary investigations about the potential production of tritiated tungsten particles by plasma abrasion of the beryllium-tungsten blankets. In particular, few is known about the capacity of these particles to absorb and further release tritium. We have been investigating both the absorption and release of tritium gas by various tungsten powders and their (radio)toxicity on cultured pulmonary cells (from 30 nm to 30 mm diameter) in collaboration with several CEA teams supporting the ITER project, in particular Drs. V. Malard (DSV) and C. Grisolia (DSM) but also Dr. F. Gensdarmes (IRSN) and academic partners Drs. D. Vrel (LSPM- Univ. Paris 13) and G. Dinescu (NILPR-Bucarest).

Tungsten particles © SCBM/CEA
Collaborations:
- CEA : Dr. C. Grisolia, Dr. V. Mallard
- Academic : Dr. D. Vrel (LSPM-Paris 13), Dr. G. Dinescu (NILPR, Bucarest), Dr. F. Gensdarmes (IRSN)
Materials
Development of chiral fluorophores for optical or photonic devices
The discovery of efficient thermally Activated Delayed Fluorescence (TADF) materials and small organic molecules enabling circularly polarized luminescence emission (CPL-SOMs) is crucial for the development of future optical and photonic devices. In TADF emitters both singlet and triplet excitons can be harvested for light emission by a reverse intersystem crossing process because there is a small energy gap between their singlet and triplet states. This property has recently motivated numerous research works because of the theoretical possibility to develop organic light emitting devices with maximum efficiency. The conception of CPL-SOMs is also a great challenge for organic chemists. Presently, only a small number of CPL-SOMs display high performance both in terms of quantum yield (φF) and luminescent dissymmetry factors (glum). Moreover, such enantiopure compounds are usually obtained after numerous synthetic steps or required enantiomeric separation through preparative HPLC restricting their application because of lack of cost-effectiveness. As a consequence, rapid, easy and flexible access to more performant CPL-SOMs is required in order to unlock their tremendous technological potential.
Our team started to work on this research area in 2015 and is currently studying the structure-activity relationships for improving CPL properties.
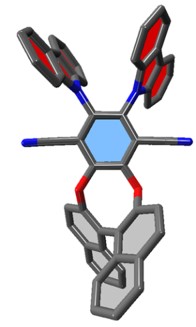

Structure of a TADF/CPL molecule and picture of an OLED
device © SCBM & LETI/CEA
Collaborations:
- CEA: Drs E. Quesnel et Dr. B. Racine
- Academic : Dr. L. Favereau (ISCR, Université de Rennes), Dr. G. Clavier (PPSM, ENS Paris-Saclay), Dr. D. Tondelier (Ecole Polytechnique), Pr. G. Muller (San Jose States University)