Alzheimer's disease affects more than a million people in France. Associating brain aging aspects and sometimes genetic predispositions, the disease results from the slow degeneration of neurons and ultimately leads to a progressive loss of memory. That degeneration starts in the hippocampus* and spreads thereafter to the rest of the brain. It is associated with the aggregation of a peptide called amyloid-β and its accumulation in the form of amyloid plaques within the brain.
Alzheimer's disease is not contagious in normal conditions. Epidemiological studies have however suggested that amyloid-β deposits can be transmitted between human patients in exceptional circumstances, such as the injection of brain-derived growth hormones, or neurosurgical procedures involving brain tissue grafts. It is thus essential to understand the aspects of the brain that, in these exceptional circumstances, enable the transmission of an amyloid pathology to a new patient. The current hypothesis is that the use of cerebral materials presenting amyloid lesions is responsible for pathology induction. Notably, in an earlier work, researchers from MIRCen's Neurodegenerative Diseases Laboratory had shown that the inoculation of brain extracts taken from human Alzheimer's disease victims into non-human primate models induced amyloid plaque formation and caused a pathology characterized by memory failure, communication difficulties, neuronal loss and decreased brain size. That work was the first illustration of the induction of clinical signs of neurodegenerative disease via the injection of human Alzheimer's disease brain extracts.
In a recent work published in Acta Neuropathologica Communications, that same team has now shown that the inoculation of brain tissue samples—determined as healthy by current standards—may also cause amyloid-β development. These latest results suggest that some transmissible elements may be undetectable by current analysis techniques. In this work, the team injected a homogenate of Alzheimer-affected human brain tissue into amyloid-β-resistant mice. Eighteen months later and despite that direct exposure to diseased tissues, the mice had apparently healthy hippocampi; they had no detectable signs of amyloid-β lesions there, or for that matter in their cortices. The researchers went on to create homogenates from those apparently healthy hippocampi and injected them into a second set of mice more susceptible to developing amyloid-β lesions. Thereafter, this second group of mice indeed developed amyloid plaques. The team's finding suggest that amyloid-β seeds from the human brain samples did persist in some stealthy form in the amyloid-β-resistant mice and even maintained their ability to provoke amyloid-β deposits once in a more receptive host.
This work underlines the need for highly diligent measures, particularly in the setting of neurosurgery, to prevent amyloid-β transmission from any cerebral tissue that is potentially affected by it, even if that tissue appears healthy under conventional analysis. In the setting of human brain surgery, numerous preventive measures have been deployed over the last few years, making the risk of this amyloid-β transmission extremely low if not inexistent.
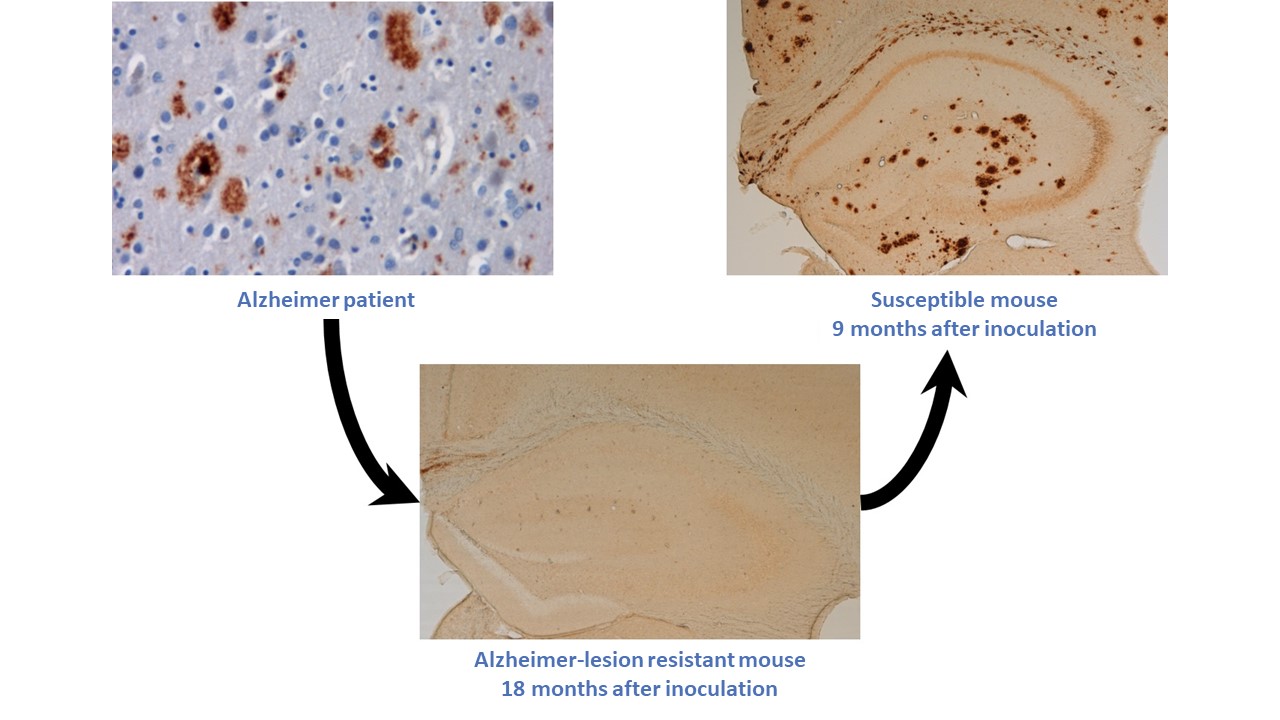
Sequential inoculation of: extracts of human brain affected by
Alzheimer's disease and presenting amyloid plaques (upper left) in a
amyloid-resistant murine model (bottom); extracts of the Alzheimer-inoculated
amyloid-resistant mouse brain (bottom) in an amyloid-susceptible murine model
(upper right), wherein some currently undetectable trace in the former provokes
the development of human-like amyloid plaques in the latter.
* The hippocampus is a cerebral structure essential for memory.
To learn more about team's work